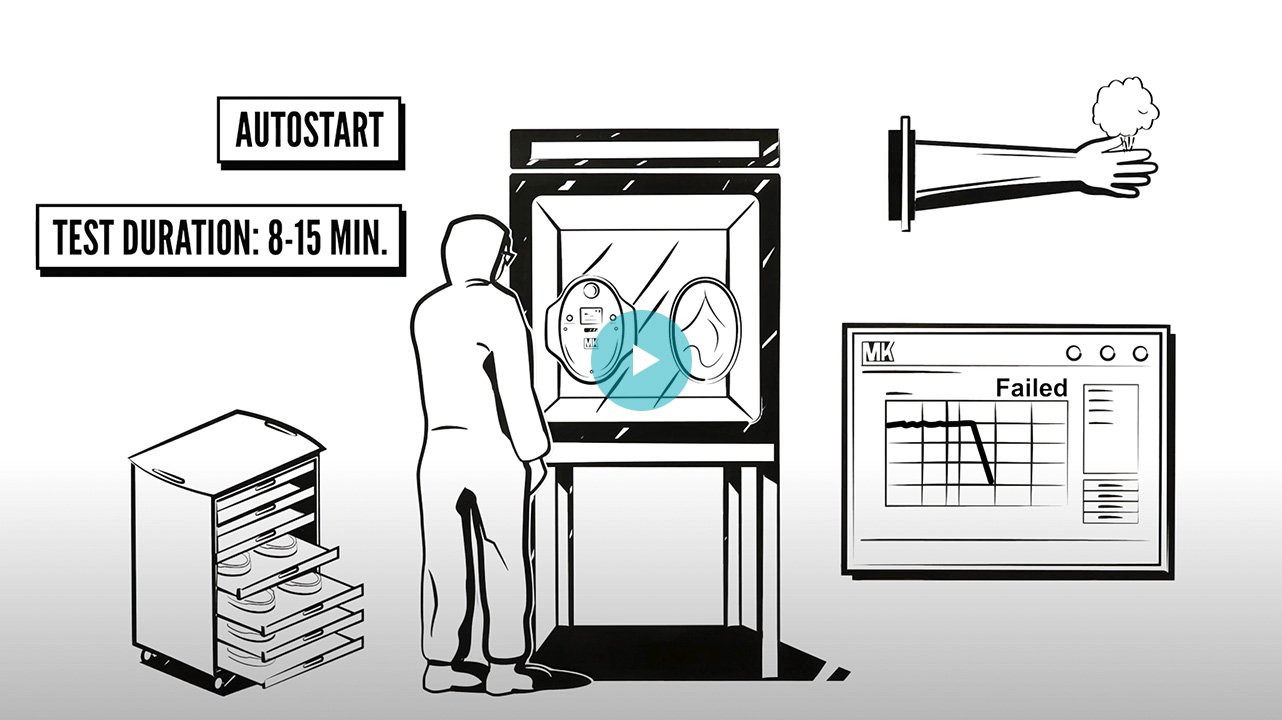
Gloves Integrity Testing (GIT) and Alpha Ports, Beta Containers Integrity Testing Devices

In the manufacture of high-potent active ingredients or in aseptic processes, safety for the operator and/or the product is essential. Therefore, in systems with isolators, the integrity of gloves and the connection of transfer systems is a very important matter. GMP Annex 1 published in August 2022 recommends regular integrity testingbefore and after each batch to ensure working conditions.
Our partner MK Versuchsanlagen is the world leader in isolator glove integrity testing systems. As Pioneers and specialists in the field, it supplies its OEM systems to the main isolator and RABS manufacturers, as well as to CMOs and the biggest pharma companies. It also has the capacity to develop customised solutions for your current isolators regardless of manufacturer. MK currently offers software solutions to manage the state of the gloves from a plant in a global manner, fully traceable via RFID and with data integrity. It is also developing solutions to test the integrity of transfer systems such as Alpha ports and Beta containers.
MK’s product range includes:
Glove Integrity Test Devices
Alpha ports Integrity Test Devices
Beta containers Integrity Test Devices
Glove Integrity Management Software and others (GITS®4)
> Glove Integrity Test Devices
> Alpha ports Integrity Test Devices
> Beta containers Integrity Test Devices
> Glove Integrity Management Software and others (GITS®4)
With MK, you have advantages which other non-specialist companies cannot offer you:
A single source supplier/system for all types of isolators, for any port type (oval, round), port material and/or glove, etc., including transfer systems (Alpha-ports and Beta-containers)
High quality product and validation package based on scientific-technical criteria. MK has an R+D department with the know-how for preparing specific recipes for any kind of requirement (pore size, cycle time, glove material, etc.).
Global system, with the possibility of scaling up towards a maximum level of automation. MK devices can be supplied to operate in stand-alone mode and adapted in the future to integrate with a management software with traceability and data integrity (GITS®4).
> A single source supplier/system for all types of isolators, for any port type (oval, round), port material and/or glove, etc., including transfer systems (Alpha-ports and Beta-containers)
> High quality product and validation package based on scientific-technical criteria. MK has an R+D department with the know-how for preparing specific recipes for any kind of requirement (pore size, cycle time, glove material, etc.)
> Global system, with the possibility of scaling up towards a maximum level of automation. MK devices can be supplied to operate in stand-alone mode and adapted in the future to integrate with a management software with traceability and data integrity (GITS®4)